Abstract
Background: Allogeneic stem cell transplantation (SCT) is the most powerful therapy to prevent relapse in poor-cytogenetics risk acute myeloid leukemia (poorAML) patients (pts) in first complete remission (CR1). For pts who lack a matched related donor (MRD), SCT from an unrelated (UD) or alternative donor is indicated. Pts with poorAML and thus at high risk of relapse can theoretically benefit the most from SCT from haploidentical donors (HaploSCT), which is an attractive option as the time required to find a well-matched UD could be inacceptable. Several recent reports show comparable outcomes between HaploSCT and transplants from UD (Piemontese S, JHO 2017; Versluis J, Blood Advances 2017). Comparative studies able to include sufficient numbers of pts with poorAML in CR1 are limited; this prompted us to compare the outcomes of HaploSCT to those of 10/10 and 9/10 HLA-matched UD in this disease category.
Methods: We retrospectively selected denovo poorAML pts in CR1 receiving T-repleted haplo (n=74), 10/10 UD (n=433) and 9/10 UD SCT (n=123) from 2007 to 2015 who were reported to the ALWP of EBMT Registry. PoorAML was defined as the presence of: complex karyotype (at least 3 structural abnormalities per clone); monosomal karyotype (1 autosomal monosomy plus 1 monosomy or structural abnormality); inv(3)/t(3;3); -5 or del(5q); -7 or abn(7q); t(v;11)(v;q23); abn(17p); t(6;9); t(9;22). Primary endpoints were leukemia-free survival (LFS) and overall survival (OS). Secondary endpoints were acute and chronic GVHD (aGVHD and cGVHD), relapse and nonrelapse mortality (NRM).
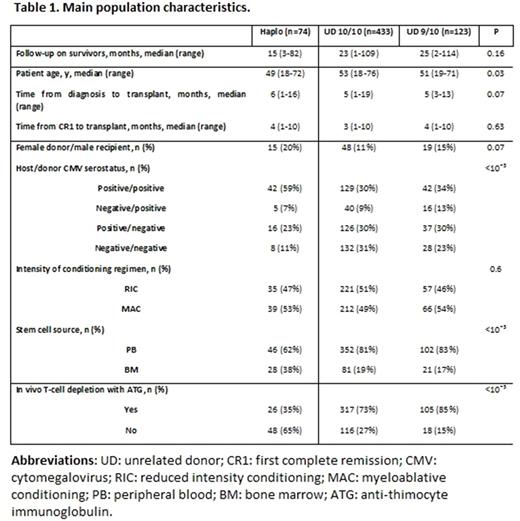
Results: Main population characteristics are depicted in Table 1. Recipients of haplo-, 10/10 UD- and 9/10 UD-SCT were comparable concerning time from diagnosis to SCT and time from CR1 to SCT. HaploSCT more likely received bone marrow as stem cell source. In-vivo T cell depletion (TCD) with ATG was most likely adopted in UD-SCT, with a conversely increased use of high-dose post-transplant Cyclophosphamide as GvHD prophylaxis backbone in HaploSCT (65% Vs 3% for 10/10 UD and 2% for 9/10 UD, p<10¯³).
LFS and OS at 2 years were not significantly different between haploSCT, 10/10 UD SCT and 9/10 UD SCT (53±12% and 59±12%, 43±5% and 50±6%, 44±9% and 50±9%, respectively, p=0.5 and p=0.5, respectively).
In Haplo-SCT, the 100-day cumulative incidence (CI) of grade≥2 aGvHD was in line with the one reported for 10/10 and 9/10 UD (33±11% for haplo, 30±4% for 10/10 UD and 34±9% for 9/10 UD, p=0.6). Likewise, the 2-y CI of cGvHD (35±12%) of HaploSCT was similar to those of 10/10 UD (36±4%) and 9/10 UD (36±9%), p=0.8. The 2-y CI of NRM was 19±8% after haploSCT, 18±4% after 10/10 UD SCT and 18±6% after 9/10 UD SCT (p=0.9). Relapse incidence was not significantly affected by donor source, with a 2-y CI of 27±9% for haploSCT, 39±5% for 10/10 UD SCT and 37±9% for 9/10 UD SCT (p=0.3).
After adjustment for centre effect, pts age, time from diagnosis to SCT, conditioning intensity, in-vivo TCD, donor/pts gender and CMV serostatus, the multivariable model showed that haploSCT recipients didn't experience worse outcomes compared to 10/10 and 9/10 UD. Indeed, compared to haploSCT (reference) the hazard ratio (HR) for LFS was 1.2 for 10/10 UD (p=0.3) and 1.2 for 9/10 UD (p=0.4). The hazards for OS in 10/10 and 9/10 UD did not differ from haplo-SCT (1.3, p 0.3 and 1.2, p 0.4, respectively). Moreover, compared to haplo, SCT from 10/10 and 9/10 UD was not associated with lower hazards for relapse (HR: 1.4, p=0.2 and 1.4, p=0.3, respectively), NRM (HR: 1, p=0.9 and 1, p=0.9, respectively), grade≥2 aGvHD (HR: 1.2, p=0.6 and 1.4, p=0.3, respectively) and cGvHD (HR: 1.2, p=0.5 and 1.3, p=0.4, respectively). The only factor associated with worse LFS and OS was pts age (for each 10-year interval: HR 1.1, p=0.02 and 1.2, p=0.001, respectively).
Conclusions: In the present series of poorAML pts transplanted in CR1, haploSCT recipients experienced comparable outcomes with respect to 10/10 and 9/10 HLA-matched UDs. This suggests that the immunotherapeutic effect of allogeneic SCT is exerted similarly across these different donor sources in this peculiar population. Therefore, in the absence of a MRD, pts with poor risk cytogenetics who have a very high risk of relapse could be allocated to haploSCT in their first remission, especially in the context of the recent improvements, which fostered an abatement of GvHD and NRM rates, historically the main detrimental factors for Haploidentical transplants.
Ciceri: GSK: Other: B-thalassemia gene therapy was developed by Fondazione Telethon and Ospedale San Raffaele and has been inlicenced by GSK that provides funding for the clinical trial, Research Funding. Mohty: Sanofi: Honoraria, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal